Translate this page into:
Brown adipose tissue – magnetic resonance imaging (MRI) appearance of these physiological fat deposits in a newborn
*Corresponding author: Hirva Manek, Department of Radiodiagnosis, Bai Jerbai Wadia Hospital for Children, Mumbai, Maharashtra, India. hirvamanek@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Manek H, Gala F. Brown adipose tissue – magnetic resonance imaging (MRI) appearance of these physiological fat deposits in a newborn. Wadia J Women Child Health. 2023;2(3):140-3. doi: 10.25259/WJWCH_36_2023
Abstract
Brown adipose tissue (BAT) is responsible for thermogenesis in neonates and infants in response to cold environment. 18F-fluorodeoxyglucose positron emission tomography is the reference for imaging of BAT, but it is a functional imaging modality and hence lacks sensitivity due to varying patient and environmental factors. Magnetic resonance imaging (MRI) lacks the use of radioactivity and ionizing radiation and can prove to be an excellent imaging modality to study the distribution of BAT. We report a neonate with sepsis whose MRI study of the neck and upper chest done to look for infective focus revealed classical distribution and signal intensity of BAT.
Keywords
Newborn
Physiological
Magnetic resonance imaging
Brown fat
Brown adipose tissue
INTRODUCTION
Adipose tissue in mammals is of two types, brown and white adipose tissue. Brown adipose tissue (BAT) is rich in mitochondria, which metabolizes fuel to produce heat through the process of non-shivering thermogenesis. Radiologically, BAT is known for its avid uptake in 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) scans, which is the current standard of reference. Research for imaging BAT involves modalities such as PET-computed tomography (CT), single-photon emission computerized tomography, magnetic resonance imaging (MRI), and magnetic resonance spectroscopy (MRS), with each modality having its own merits and demerits.[1] We report a case of a neonate with sepsis showing characteristic distribution and signal characteristics of BAT on conventional MRI.
CASE REPORT
A male child, born full-term out of a non-consanguineous marriage, had a weak cry at birth and hypoglycemic seizures on the second day of life. The blood glucose at the time of the convulsion was 46 mg/dL. On day of life 7, he was referred to our hospital for treatment of sepsis. Serum C-reactive protein levels were high – 101 mg/L with significantly raised total white blood cell counts. A sonogram of the chest and abdomen to look for any focus of infection was unremarkable except for a few subcentimeter-sized hypoechoic oval-shaped lymph nodes with preserved fatty hilum in bilateral axilla and inguinal regions. These appeared reactive but an MRI of the neck and upper chest was done to rule out any infective focus. The images showed bilateral symmetrical multilocular, homogenous altered signal intensity sheet-like areas in bilateral supraclavicular and infraclavicular spaces, axilla, paraspinal, and interscapular regions [Figures 1-4]. This soft-tissue appeared isointense to muscle on T1 images, hyperintense to muscle, and hypointense to subcutaneous fat on T2 images and short tau inversion recovery images (STIR) and showed homogenous post-contrast enhancement. A similar intensity soft tissue deposit was seen adjacent to the right pericardium [Figure 5]. The symmetry, distribution, and signal intensity of these areas suggested these to be the physiological BAT deposits in neonates. No focus of infection was seen on the MRI scan.
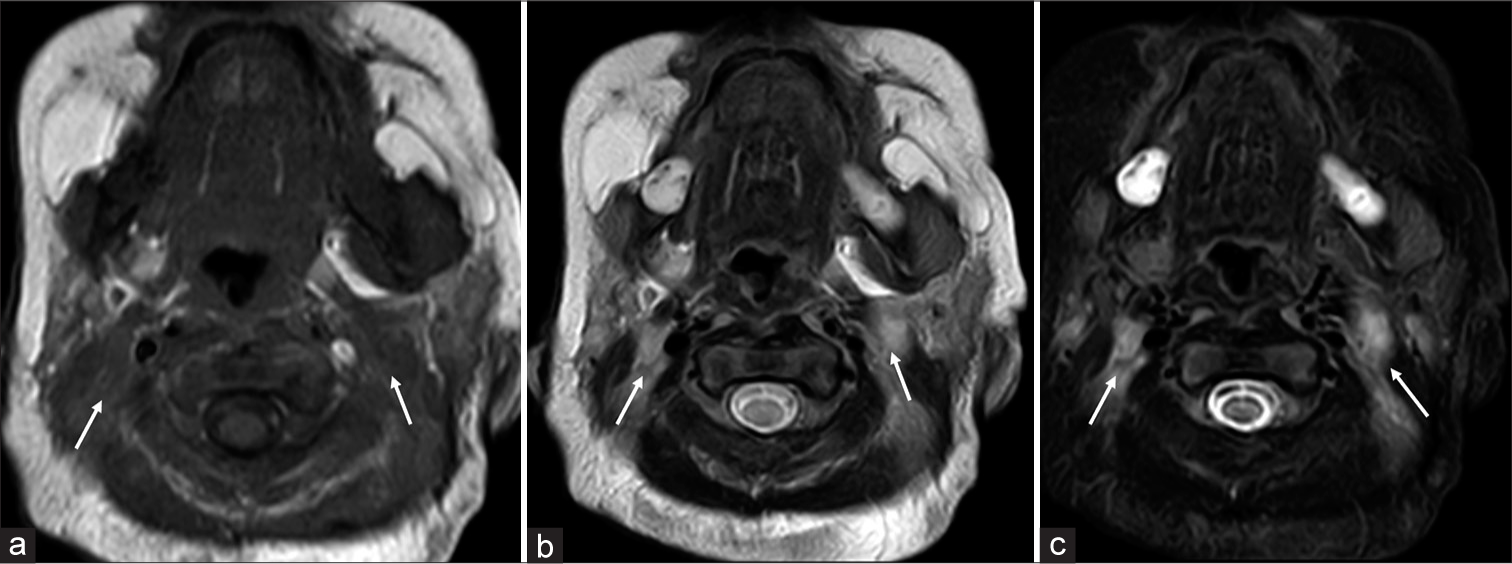
- Axial images at the level of neck: (a) T1W image shows isointense sheet like tissue in neck on both sides, deep to the sternocleidomastoid and lateral to the carotid and jugular vessels (white arrows). (b) On T2W image this soft tissue appears hyper intense (white arrows). (c) Hyper intensity of the soft tissue on short tau inversion recovery (STIR) image as well (white arrows).
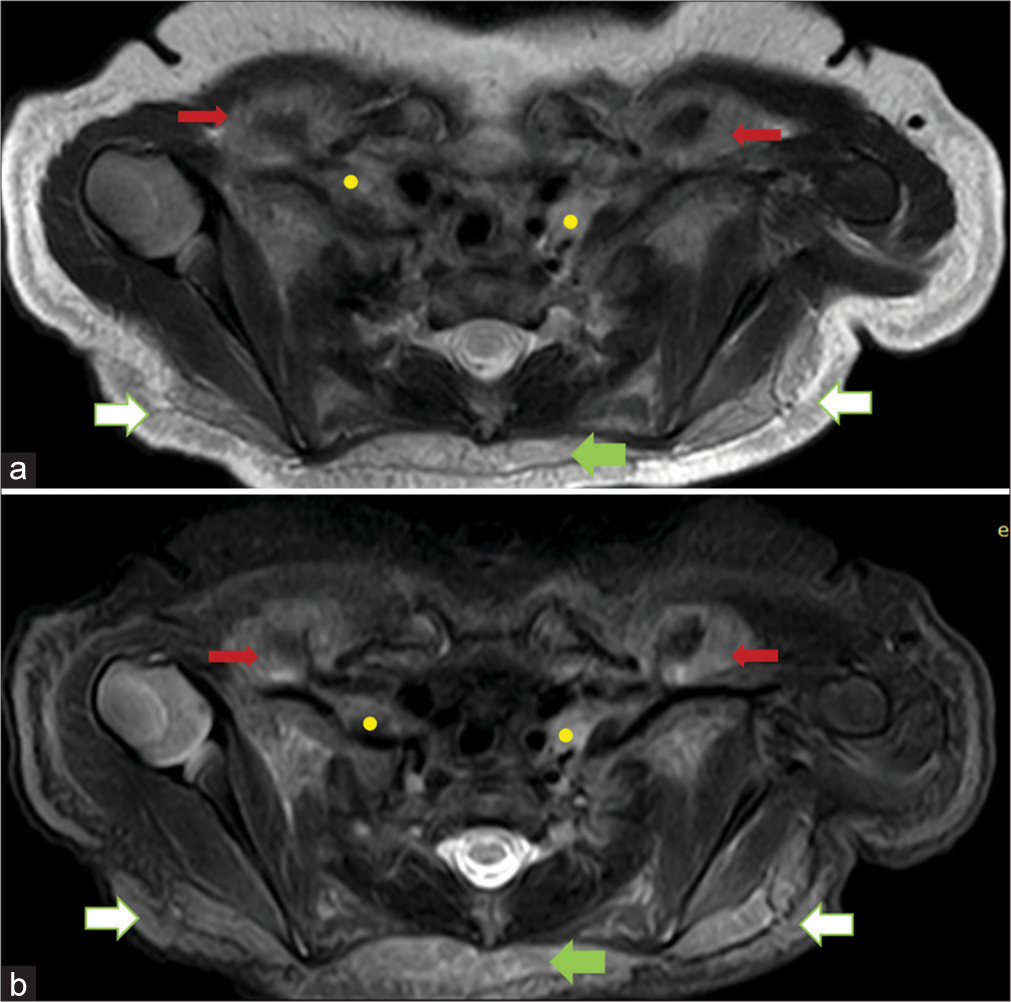
- Axial images at the level of thoracic outlet: (a) T2 intermediate soft tissue in the interscapular region (green arrow), supraclavicular (yellow dots) and infraclavicular spaces (red arrows) and in the subcutaneous tissue of back (white arrows). (b) Short tau inversion recovery (STIR) hyperintensity in the same areas, interscapular region (green arrow), supraclavicular space (yellow dots) and infraclavicular spaces (red arrows) and in the subcutaneous tissue of the back (white arrows).
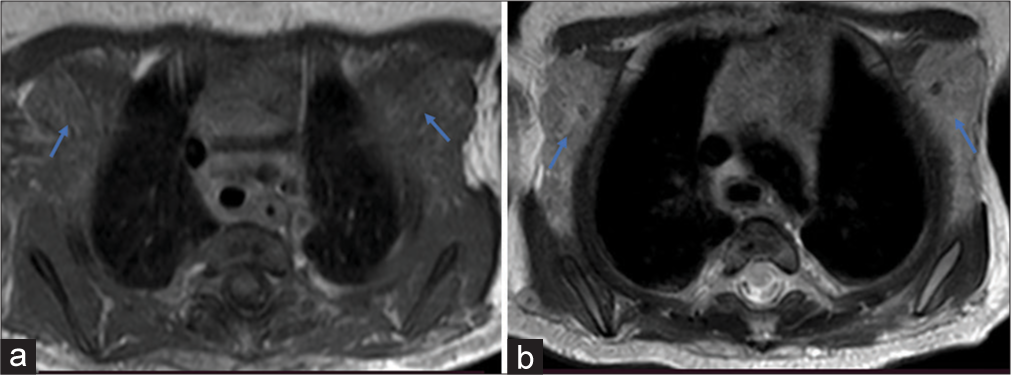
- Axial images at the level of axilla: (a) T1W image shows isointense soft tissue in both axilla (blue arrows). (b) T2W image shows lobulated hyper intense soft tissue in both axilla (blue arrows).
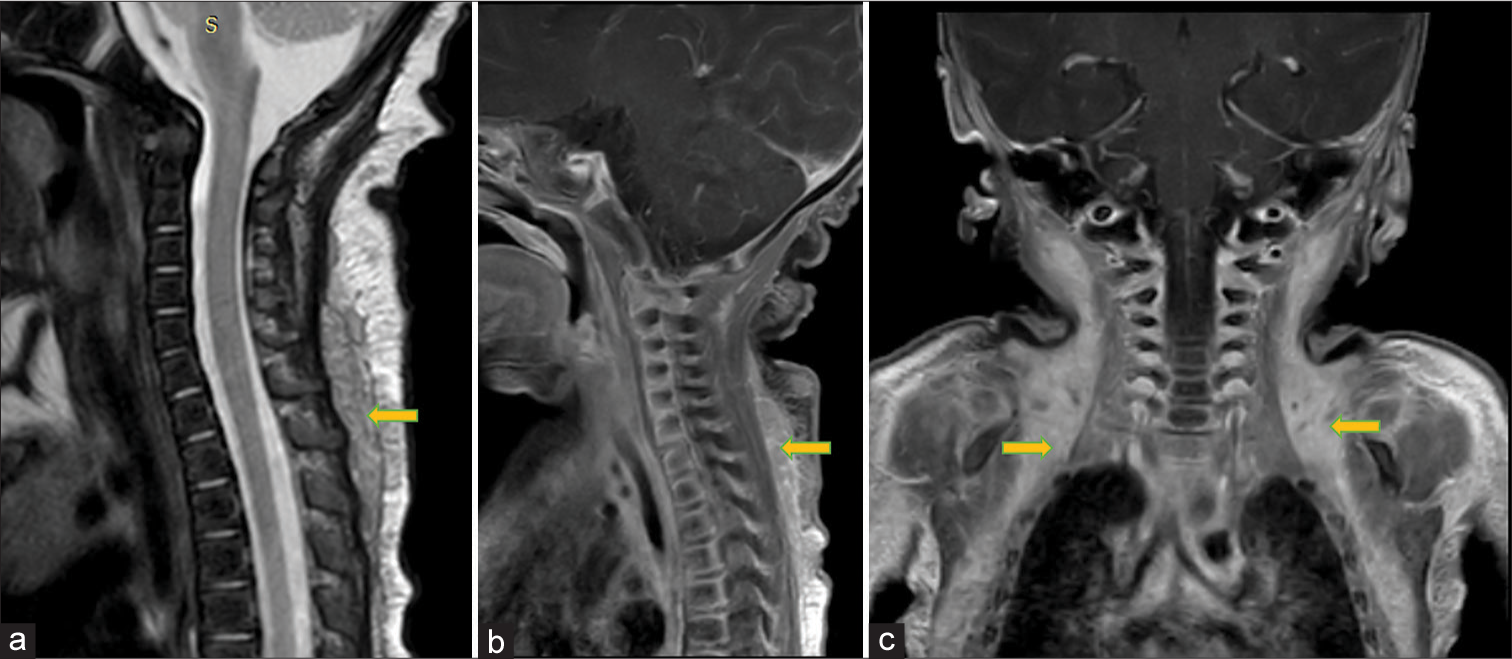
- Sagittal and coronal images of cervical spine: (a) Sagittal T2W image shows intermediate signal intensity soft tissue in the subcutaneous plane of back in midline, posterior to paraspinal muscles (yellow arrow). (b) Sagittal post contrast fat saturated T1W image shows enhancement of this mid line subcutaneous tissue (yellow arrow). (c) Coronal post contrast fat saturated T1W image shows homogeneously enhancing tissue in the neck, axilla and chest wall on both sides (yellow arrows).
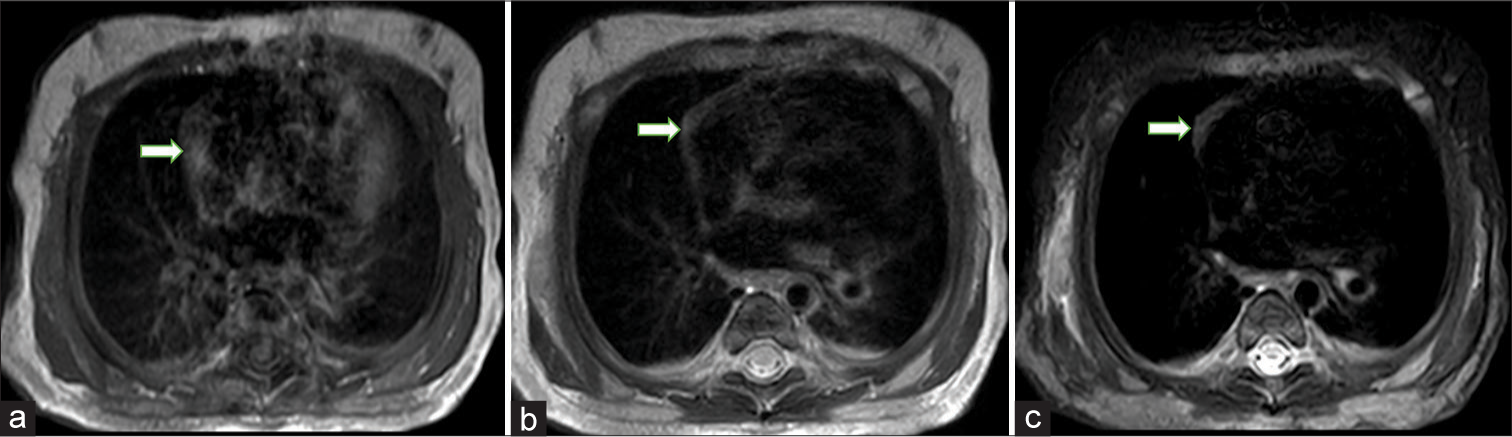
- Axial images at the level of mediastinum: (a) T1W image shows small iso intense mound of soft tissue (white arrow) to the right of heart (b) It appears intermediate to slightly hyper intense on T2W image (white arrow). (c) Short tau inversion recovery (STIR) image also shows hyper intensity of the tissue (white arrow).
DISCUSSION
BAT, known as the hibernating gland in older times, is seen in hibernating as well as many non-hibernating mammals such as cats and rats. It is rich in mitochondria, which gives its characteristic brown color. It is responsible for thermoregulation by non-shivering thermogenesis in cold conditions. At the molecular level, there are two types of thermogenic adipose tissue – brown and beige adipose tissue. These two cells have different cell lineages and express different selective marker genes. The sensation of cold from the peripheral receptors reaches the hypothalamus which, in turn, stimulates the sympathetic nervous system supplying the BAT. Grossly, BAT is arranged as multilocular deposits with intervening connective tissue septa. A connective tissue layer also separates it from the adjacent white adipose tissue.[2]
BAT in humans is anatomically divided into visceral and subcutaneous fat. The visceral fat is seen in the (1) perivascular space around the major vessels of the neck, aorta, epicardial coronary arteries, internal mammary, and intercostal arteries, (2) visceral BAT in the mediastinum around the heart, greater omentum, and in the transverse mesocolon, and (3) around the solid organs such as kidneys, adrenals, liver, and the splenic hilum. The subcutaneous BAT is seen in the axilla, neck, supraclavicular fossa, interscapular, paravertebral spaces, anterior abdominal wall, and in the inguinal regions. The BAT deposits at all these sites are seen in infants and neonates and decrease with increasing age.[3]
In ancient times, when BAT was discovered in mammals, including neonates and adults, it was discovered and studied in autopsy specimens. The advent of imaging has made it possible to study BAT in vivo, both structurally and functionally. 18F-FDG PET is the reference standard as the BAT shows avid uptake of the metabolic tracer. However, not all BAT deposits take up the tracer due to inactivity. Furthermore, PET needs patient exposure to hypothermia for optimal tracer uptake, needs injection of the radioactive material, and is useful only in patients with oncological conditions as PET study is used in these patients for their screening and treatment follow-up. It is also not ethical to expose healthy individuals to radioactive study for analysis of the BAT. This lacuna in imaging of BAT is overcome with the help of MRI as an imaging modality. Furthermore, MRS can help in molecular quantification of BAT.
MRI studies of BAT suggest that it has a low T2 relaxation time and intermediate fat fraction. It appears isointense to slightly hyperintense to muscle on T1 images and intermediate to hyperintense on T2 images with partial suppression on fat sat images.[1,2,4] The soft tissue in our case showed similar signal characteristics. The multilocular appearance seen in our case is consistent with the glandular appearance of the interscapular brown fat described by Hatai.[5] The drawback of this case was that we did not confirm the tissue on pathology due to ethical reasons, and MRS was not performed for molecular analysis.
There was no other differential in this case. But if the soft-tissue deposits had been mass-like, encapsulated with or without fat signal on MRI, a suspicion of a mass lesion such as hibernoma, lipoma, or rhabdomyoma or malignancy such as rhabdomyosarcoma would have been likely. It is imperative to be careful of these features to differentiate between the physiological and pathological soft tissue, as the latter requires confirmatory diagnosis and definitive management.
CONCLUSION
BAT deposits are physiological fat deposits seen at characteristic sites on imaging, and their symmetry and signal intensity on MRI are characteristic. These are responsible for non-shivering thermogenesis in neonates and infants and these deposits decrease with increasing age. MRI is a non-invasive modality for imaging these fat deposits as it does not require patient preparation or drug injection.
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship
Nil.
References
- Imaging brown adipose tissue using magnetic resonance: A promising future? Radiology. 2021;299:407-8.
- [CrossRef] [PubMed] [Google Scholar]
- Brown adipose tissue in human infants. Brown adipose tissue 2019:107-23.
- [CrossRef] [PubMed] [Google Scholar]
- Anatomical locations of human brown adipose tissue: Functional relevance and implications in obesity and Type 2 diabetes. Diabetes. 2013;62:1783-90.
- [CrossRef] [PubMed] [Google Scholar]
- Brown adipose tissue in a newborn. Proc (Bayl Univ Med Cent). 2008;21:328-30.
- [CrossRef] [PubMed] [Google Scholar]
- On the presence in human embryos of an interscapular gland corresponding to the so-called hibernating gland of lower mammals. Anat Anz. 1902;21:369-73.
- [Google Scholar]







