Translate this page into:
Leukocyte adhesion defect: Clinical correlation with integrin expression and genetics
*Corresponding author: Dr. Mukesh M. Desai, Head, Division of Immunology, Department of Pediatrics, Bai Jerbai Wadia Hospital for Children, Mumbai, Maharashtra, India. desai.mm18007@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Iyengar VV, Taur P, Gowri V, Chougule A, Kambli P, Madkaikar M, et al. Leukocyte adhesion defect: Clinical correlation with integrin expression and genetics. Wadia J Women Child Health 2022;1(1):3-7.
Abstract
Objectives:
Leucocyte adhesion defect 1 (LAD1) is an autosomal recessive defect in integrin expression. In this study we present a case series of 19 children with LAD1. We analysed their clinical and laboratory data and correlated it with integrin expression and underlying genetic defect.
Material and Methods:
Retrospective case record study of all children diagnosed with LAD1 between 2012 and 2021.
Results:
Ten females and 9 males were included. Eight cases were born of consanguineous marriage. 78% (15/19) had disease onset within one year of life and 8 of them were neonates. Neonates with LAD1 commonly presented with omphalitis, late-onset sepsis and respiratory infections (RTI). Most common organism isolated from blood was pseudomonas (4/8). CD11/CD18-expression ranged from severely reduced (6/8) to moderately reduced (2/8) but disease was uniformly fatal in the 7/8 patients who could not undergo hematopoietic stem cell transplant (HSCT). Seven children presented between 1month and 1year of age with RTI, bacterial sepsis and soft tissue abscess. Four expired, 1 survived after HSCT and 2 were alive at last follow up. CD11/CD18-expression ranged between 0-1.6% except in 2 cases. One child with 11%-expression is alive without transplant. Another child had 0%-CD11 but 17%-CD18-expression with very low MFI (mean florescence intensity). She succumbed at 20 months of age with perianal abscess, sepsis and RTI but prior to this fatal episode she developed autoimmune cutaneous lesions with lymphocytic infiltration. Four children presented beyond infancy. Median age at onset of symptom was 2 years with skin abscess, oral ulcers, severe periodontitis with tooth loss and pyogenic arthritis. Three of them survived without HSCT, 1 was alive at last follow up. CD11/CD18-expression ranged from 20-32% in these children. Details of cord separation was available in 17 cases and it was delayed (beyond 2 weeks of life) in 9 children. Of the 17 cases with genetic analysis of ITGB2 gene, 16 had a homozygous and 1 had compound heterozygous variant. (nonsense: 2; splice site variants:5; 8: missense variants; 2: deletion).
Conclusion:
Disease onset less than 1 year age was fatal, irrespective of CD11/CD18 expression. Integrin expression levels beyond 20% and splice site mutation were associated with 100% survival without HSCT but were not event free. Pseudomonas sepsis is the commonest infection in LAD1 neonates. Between 1month to 1year, RTI and gram-negative sepsis dominated and beyond 1st-year skin/perianal abscess was common. Delayed separation of cord was present only in 52% of cases.
Keywords
Leucocyte adhesion defect
Omphalitis
CD11
CD18
Inborn errors of immunity
Phagocytic defect
INTRODUCTION
Inborn errors of immunity (IEI) are a group of genetic disorders that affect distinct components of the innate and adaptive arms of immune system. Leukocyte adhesion defect 1 (LAD1) is a rare disorder causing abnormality in leucocyte adhesion and migration occurring due to autosomal recessive mutations in ITGB2 gene encoding β2 integrin (CD18) which is essential for adhesion of leucocytes to endothelium and transmigration to site of inflammation.[1] First described in 1979,[2] LAD 1 leads to severe, recurrent, life-threatening bacterial infections accompanied by poor wound healing and high mortality. Here we present a comprehensive retrospective report of 19 patients diagnosed at our centre along with their clinical features, integrin expression and genetic mutations.
MATERIAL AND METHODS
This was a retrospective study of records of 19 patients, proven to have LAD1 at our hospital between 2012-2021. Diagnosis was based on demonstrating absence or reduced expression of CD11/CD18 by flow cytometry or genetic mutations in ITGB2 gene. The clinical details including age at onset, sex, family history, consanguinity in parents, infection profile, presence or absence of delayed cord separation (beyond 2 weeks after birth), medication history, growth parameters and clinical examination features were recorded. Laboratory data included information about isolated organisms, total leucocyte count, and absolute neutrophil count. Flow cytometric analysis for integrin expression was performed with a flow cytometer to determine the CD18, CD11a, & CD11c expression on cells from whole blood. Results were displayed using percentage of cells containing each CD and MFI. Genetic diagnosis of LAD 1 by exome sequencing followed by Sanger confirmation of the variant in ITGB2 was done.
RESULTS
There were 10 female and 9 male patients, out of whom 8 were born of consanguineous marriage. A positive family history of early sibling deaths was found in 6 of them. Fourteen of these cases have been partly reported previously as part of multicentric case series.[3]
Fifteen children had onset of symptoms in the first year of life out of which 8 were symptomatic in neonatal period [Figure 1].
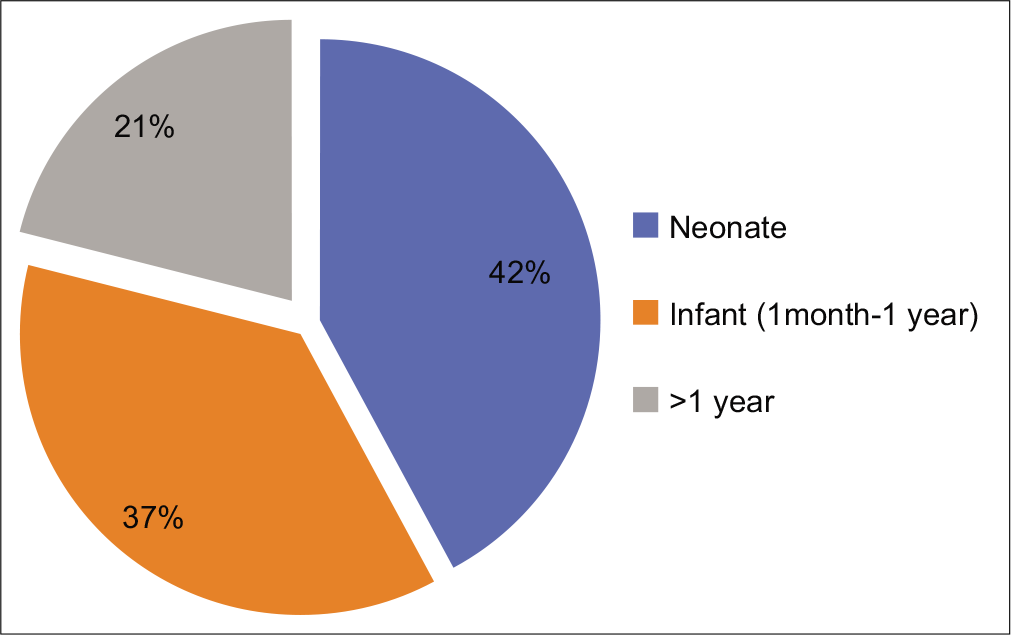
- Age at onset.
Details of separation of cord was available in 17 cases. Umbilical cord fall was delayed in 9/17 (52%) patients. Cord fall was delayed in 5/8 children with neonatal onset of symptoms and in 4/7 with infantile onset of symptoms. None of the 4 children with onset of symptoms more than1 year of age had delayed separation of cord fall.
Omphalitis (5/8), late onset neonatal sepsis (5/8) and RTI (5/8) were the common infections in neonates. In children between 1 month to 1 year of life, RTI (4/7), systemic sepsis (4/7) (Pseudomonas, Klebsiella, Staphylococcus) and soft tissue abscesses (4/7) were common. Intestinal perforation, pyogenic arthritis and CNS infection was seen in one child each. [Figure 2] shows the common organisms grown in this cohort of children with sepsis and LAD1.
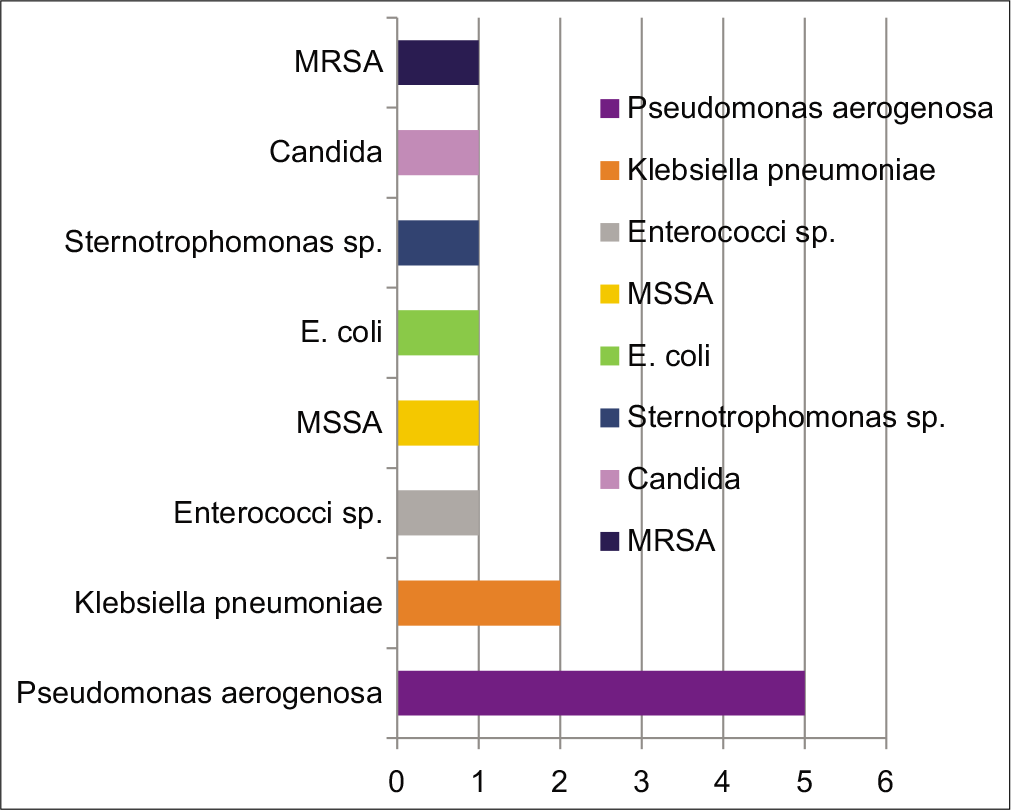
- Organisms commonly found causing sepsis in children with LAD1. MSSA: Methicillin sensitive Staphylococcus aureus, E. coli: Escherichia coli, MRSA: Methicillin resistant Staphylococcus aureus.
Beyond first year of life, skin abscess (3/4), oral ulcers (2/4), severe periodontitis with tooth loss (1/4) [Figure 3] and pyogenic arthritis (1/4) were seen.
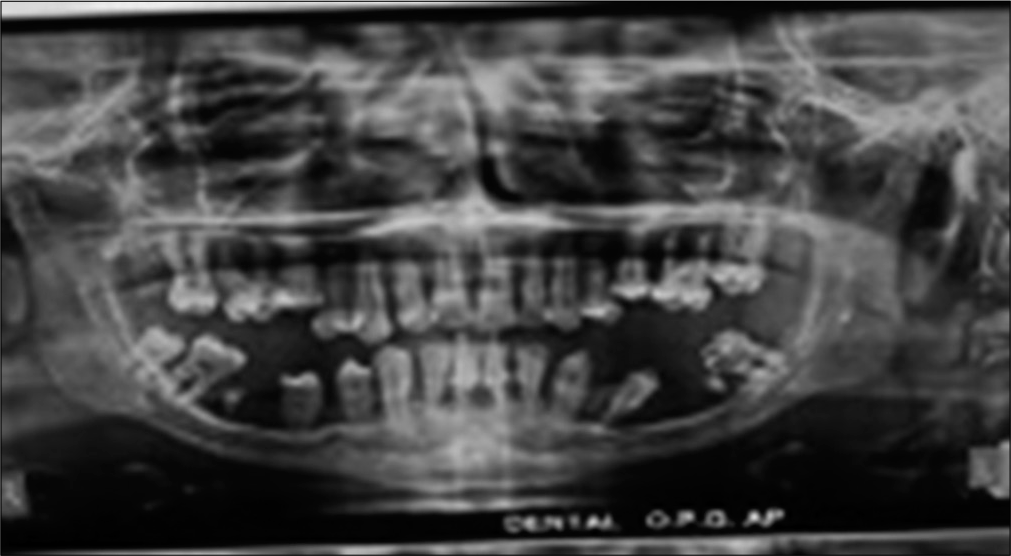
- Severe periodontitis and loss of secondary dentition in a case of mild LAD.
All children had neutrophilic leucocytosis. Total leucocyte counts ranged from 12,800 cells/mm3 to 108,300 cells/mm3 with neutrophils constituting 53% to 92% of the leucocyte population. All neonates had integrin expression ranging from 0% to 3% [Table 1]. Among infants between 1 month and 1 year of age all children had integrin expression between 0 and 1.6% [Table 1] except in 2, one of whom had expression of 11% and is alive without a transplant and another had discrepancy between CD11 and CD18 expression. This female child had CD 11 expression of 0% while CD18 expression was 17% with very low MFI. She had recurrent episodes of eczematoid skin lesions with lymphocytic infiltration of skin [Figure 4] and succumbed at 20 months of life with perianal abscess (expressing pus after granulocyte transfusion), sepsis and LRTI. In children more than 1 year of age, integrin expression was >20% [Table 1].
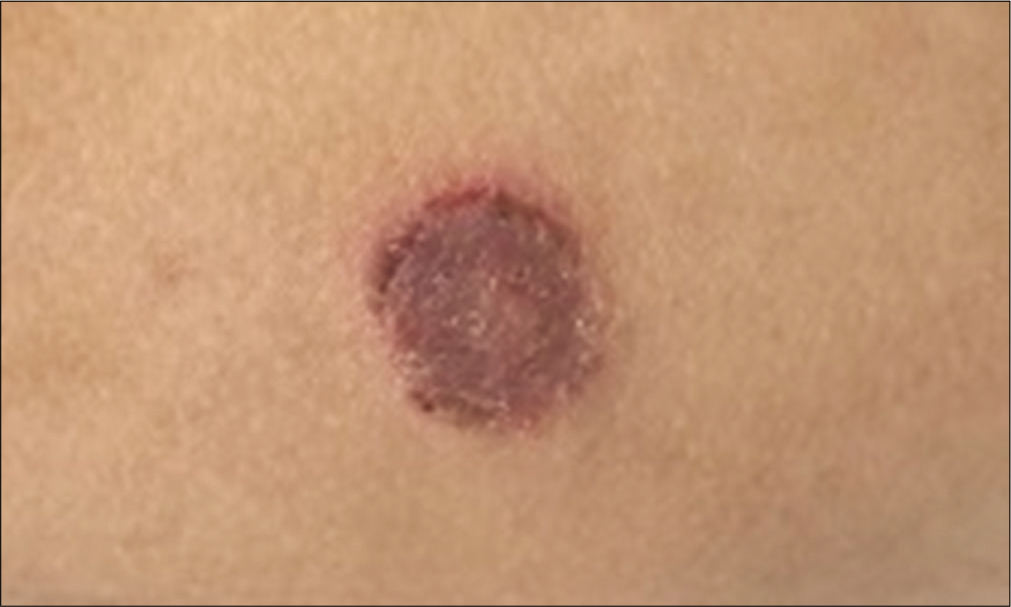
- Eczematoid skin lesions in a child with LAD1.
| Case | CD18 expression (%) | CD11 expressio n (%) | Type of mutation | Location on gene | Mutation | Outcome |
|---|---|---|---|---|---|---|
| N1 | 0.90 | 0.90 | Homozygous, missense | Exon 6 | c.533C>T p.Pro 178Leu (reported) | Expired |
| N2 | 2 | 0 | Homozygous, deletion | Exon 5 | c.461_479del p. L154Pfs*21 (novel) | Expired |
| N3 | 0.50 | NA | NA | Expired | ||
| N4 | 0 | 0 | NA | Expired | ||
| N5 | 0 | 0 | Homozygous, missense | Exon 7 | c.817G>A p. Gly273Arg (reported) | Expired |
| N6 | 0 | 0 | Homozygous, nonsense | Exon 6 | c.658G>T p. Glu220X (novel) | Expired |
| N7 | 0 | 0 | Compound | Exon 6 | c.562C>T, p. Arg188Ter and | Expired |
| heterozygous (Missense + Nonsense) | c.533C>T, p. Pro178Leu (reported) | |||||
| N8 | 3 | 3 | Homozygous, Missense | Exon9 | c920T>G p. Leu307Arg (novel) | Alive post |
| BMT | ||||||
| I1 | 11 | 11 | Homozygous, deletion | Exon8 | c.977_979delTGG p. Val327del (novel) | Alive |
| I2 | 1.60 | 1.60 | Homozygous, missense | Exon5 | c.382G>T pAsp128Tyr (reported) | Expired |
| I3 | 0 | 17 | Homozygous, Missense | Exon8 | c.850G>A p. Gly284Ser (reported) | Expired |
| I4 | 0 | 0 | Homozygous, nonsense | Exon 4 | c.322C>T p. Arg108Ter (novel) | Expired |
| I5 | 0 | 0 | Homozygous, missense | Exon 8 | c.962C>A p. Ala321Glu (novel) | Expired |
| I6 | NA | NA | Homozygous, splice site | Intron 6 | c.741G>A IVS6-1 G>A (reported) | Alive post |
| BMT | ||||||
| I7 | NA | NA | Homozygous, splice site | Intron 7 | c.897+1 G>A IVS7+1G>A (reported) | Alive |
| C1 | 20 | 50 | Homozygous, splice site | Intron10 | c.1224+4A>G IVS10+4A>G (novel) | Alive |
| C2 | NA | NA | Homozygous, missense | Exon7 | c.809C>T p.Ala270Val (reported) | Alive |
| C3 | NA | NA | Homozygous, splice site | Intron 6 | c.741G>A IVS6-1 G>A (reported) | Alive post |
| BMT | ||||||
| C4 | 32 | 28 | Homozygous, splice site | Intron 4 | c.332-2A>G IVS 4-2 (novel) | Alive |
Most common genetic mutations in ITGB2 gene were missense and splice site homozygous variants [Figure 5, Table 1].
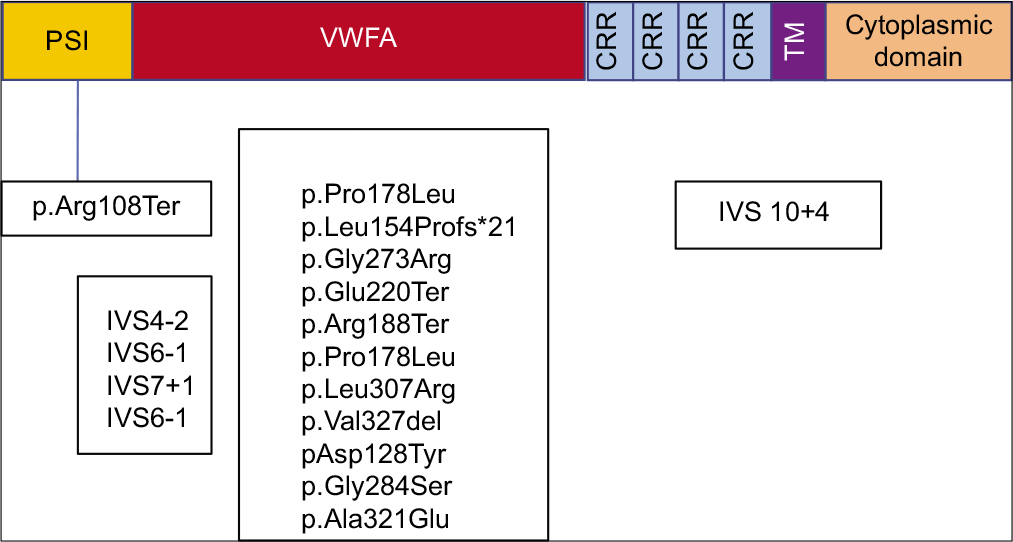
- β2 integrin protein with genetic variants. PSI: Plexinsemaphorin-integrin, VWFA: Von Willebrand factor A, CR: Cysteine rich repeats, TM: Transmembrane. (Colour code blue: Splice variants, green: Missense, red: Nonsense variants, violet: deletion).
Two children received granulocyte transfusion, one of whom developed respiratory decompensation post the transfusion.
The disease was fatal in all but one with neonatal onset and in majority with infantile onset (between 1 month and 1 year). All children (4/4) with onset more than 1 year age are alive. All children (5/5) with splice site variants are alive.
DISCUSSION
Leucocyte adhesion defect is a phagocyte disorder characterized by inability of cells to transmigrate across vascular spaces into tissues due to abnormality in the integrin expression. Integrins are required for strong adhesion of fast-moving blood cells onto endothelium.[4] Phagocytes, predominantly neutrophils, are unable to egress from circulation and hence manifest as lesions that have absence of pus, that do not localize and do not heal leading to persistent inflammation and spread of infection. These neutrophils are retained in circulation leading to leucocytosis [Figure 6].[5]
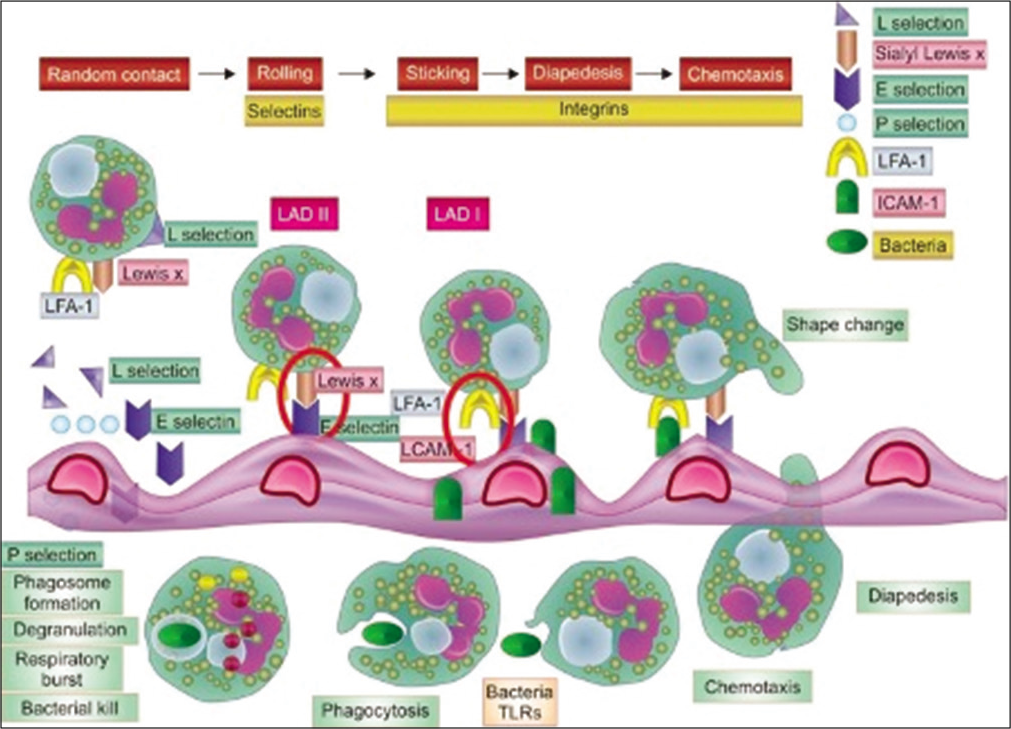
- Pathology of LAD: Defect lies in integrin leading to poor adhesion on endothelium, diapedesis and chemotaxis.
Integrin expression of <2% is considered as severe LAD1, 2–30% expression is moderate and expression beyond 30% as mild LAD1.[6] In our study, 42% of cases were born of consanguineous union and 31% had a history of early sibling death, suggestive of an autosomal recessive inheritance. Delayed separation of umbilical cord was noted in 52% of cases which is comparable to previous studies.[7] Fifteen (79%) of our cases presented in the 1st year of life with 53% of them presenting in the first month of life which was similar to other studies.[7]
Previous studies have shown that in addition to infections involving skin, gingiva and lungs,[7] other infections like life-threatening septicaemia, deep seated abscesses including in brain[8] and osteomyelitis[9] are also seen. Features of sepsis was among the commonest presenting symptoms in our patients who had disease onset <1 year of age. Pyogenic arthritis and CNS infection were also seen in one patient each. Multiple gram positive and gram negative bacteria cause infections in LAD1 patients.[7] In our cohort Pseudomonas aeruginosa was the commonest organism seen. Recent study[10] has proven the role of CD18 integrins in internalization by phagocytes of motile and nonmotile P. aeruginosa. Host phagocytosis is the principal pathway for control of pseudomonas infection and this forms the basis of increased susceptibility to pseudomonas sepsis in these patients. Poor wound healing and persistent microbe induced hyperinflammation lead to non-healing perianal ulcers and severe periodontitis.[11] High local IL23 and IL17 lead to bone resorption[12] and teeth loss as seen in one of our patients shown in [Figure 3]. Autoimmunity has been described in patients of LAD1 including renal, intestinal autoimmune diseases like chronic enterocolitis and Crohn-like colitis. One of our patients had skin lesions which on biopsy showed lymphocyte infiltration [Figure 4]. Skin autoimmunity has not been reported with LAD1.
Highlighting the importance of evaluating both CD11 and CD18 expression is the discrepancy in expression seen in one of our patients. She had 17% expression of CD18 with low MFI while her CD11 expression was 0%. She presented in the first year of life with pneumonia and thereafter had no infections but recurrent eczematoid skin lesions. She finally succumbed to perianal abscess and pneumonia at the age of 20 months. Previous studies have found variable and sometimes normal expression of CD18 and consider reduced CD11 expression to be a better indicator of LAD1.[13,14]
In this study, genetic testing could be done in 17/19 cases. 8 variants were novel [Table 1]. Similar to previous studies the most common genetic variants identified were missense mutations.[14] All children with splice mutations are alive and they presented late in life (age at presentation ranging from 6 months to 2 years). Integrin expression was available for two children with splice variants and they were >30% (mild LAD1).
Bone marrow transplant is the only curative therapy for LAD1. Three children have been transplanted in our cohort and all of them are alive. All of the 8 children with neonatal onset of symptoms succumbed except one child who underwent hematopoietic stem cell transplant (HSCT). All children <1 year age at onset also succumbed except 3, 2 of whom underwent HSCT. Although antibiotic prophylaxis is recommended for those with mild to moderate LAD1, the age at onset and severity of symptoms should guide the decision for transplant. One-time CD18 expression alone does not reflect the severity of LAD.[13]
CONCLUSION
This study highlights the fact that all children with age of onset of symptoms of <1 year age should be offered bone marrow transplant irrespective of CD11/CD18 expression as the disease is fatal in majority of them. CD11 expression and MFI correlate better with survival. Integrin expression levels beyond 20% and genetic mutation showing splice site variants were associated with 100% survival without transplant but had recurrent illnesses. Hence decision to transplant should be individualized in these patients. Pseudomonas sepsis is the commonest infection in LAD1 neonates and infants. Between 1 month-1 year of age respiratory infections and gram-negative sepsis dominated and beyond 1st-year, skin and perianal abscess were common. Delayed cord fall was present only in 52% of patients hence it should no longer be considered essential for diagnosis.
Acknowledgments
We would like to acknowledge Dr. Zinet Currimbhoy who started the immunology department at B. J. Wadia Hospital for Children in early 2000s and continues to support and mentor the department. Dr. Ambreen Pandrowala from Bai Jerbai Wadia hospital for children for patient management.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Leucocyte adhesion deficiency. Arch Dis Childhood. 1993;69:463-6.
- [CrossRef] [PubMed] [Google Scholar]
- Delayed separation of the umbilical cord, widespread infections, and defective neutrophil mobility. Lancet. 1979;313:1099-101.
- [CrossRef] [Google Scholar]
- Clinical and genetic spectrum of a large cohort of patients with leukocyte adhesion deficiency Type 1 and 3: A multicentric study from India. Front Immunol. 2020;11:612703.
- [CrossRef] [PubMed] [Google Scholar]
- Deficiency of a surface membrane glycoprotein (mo1) in man. J Clin Invest. 1984;73:153-9.
- [CrossRef] [PubMed] [Google Scholar]
- Leukocyte adhesion deficiencies. Ann N Y Acad Sci. 2012;1250:50-5.
- [CrossRef] [PubMed] [Google Scholar]
- Leukocyte adhesion deficiency-i: A comprehensive review of all published cases. J Allergy Clin Immunol. 2018;6:1418-20.e10.
- [CrossRef] [PubMed] [Google Scholar]
- Leucocyte adhesion deficiency-a multicentre national experience. Eur J Clin Invest. 2019;49:e13047.
- [CrossRef] [PubMed] [Google Scholar]
- Brain abscess in a child with leukocyte adhesion defect: An unusual association. J Clini Immunol. 2016;36:624-6.
- [CrossRef] [PubMed] [Google Scholar]
- Osteomyelitis in leukocyte adhesion deficiency Type 1 syndrome. J Infect Dev Ctries. 2010;4:175-8.
- [CrossRef] [PubMed] [Google Scholar]
- Distinct contributions of cd18 integrins for binding and phagocytic internalization of Pseudomonas aeruginosa. Infect Immun. 2020;88:e00011-20.
- [CrossRef] [PubMed] [Google Scholar]
- Subgingival microbial communities in leukocyte adhesion deficiency and their relationship with local immunopathology. PLoS Pathog. 2015;11:e1004698.
- [CrossRef] [PubMed] [Google Scholar]
- Interleukin-12 and interleukin-23 blockade in leukocyte adhesion deficiency Type 1. N Engl J Med. 2017;376:1141-6.
- [CrossRef] [PubMed] [Google Scholar]
- Highlighting the problematic reliance on cd18 for diagnosing leukocyte adhesion deficiency Type 1. Immunol Res. 2016;64:476-82.
- [CrossRef] [PubMed] [Google Scholar]
- Hematologically important mutations: Leukocyte adhesion deficiency (first update) Blood Cells Mol Dis. 2012;48:53-61.
- [CrossRef] [PubMed] [Google Scholar]






