Translate this page into:
Cutaneous skeletal hypophosphatemia syndrome
*Corresponding author: S. Rao Chandrashekhar, Professor, Division of Pediatric Endocrinology, Department of Pediatrics, Bai Jerbai Wadia Hospital for Children, Mumbai, Maharashtra, India. c_sudha@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Keshwani R, Chandrashekhar SR, Mehta A, Patil A. Cutaneous skeletal hypophosphatemia syndrome. Wadia J Women Child Health 2022;1(1):36-7.
Described here is a four year three months old girl, born of third-degree consanguineous marriage, birth and developmentally normal, presenting with bowing of legs since one year of age and multiple hairy birth marks. She was short (Height- 85.1 cm, -4 SDS) with features of rickets, genu varus, tibia vara with an intercondylar distance of 10 cm [Figure 1-a], multiple hairy nevi all over the body [Figure 1-b and d] along with a verrucous lesion on scalp [Figure 1-c]. Investigations revealed S. Calcium- 10.1 mg% (8.8-10.8), Phosphorous - 3.4 mg% (3.2-5.5), Alkaline Phosphatase-726 U/L (122-335), Tubular resorption of phosphorus - 93% (85-95%), TmP/GFR- 3.16 (3.25-5.1), S. 25(OH)D3- 9.5 ng/mL, S. FGF-23- 6300 RU/mL (0-150). A 68GaDOTATATE PET/CT did not reveal any focus of abnormal tracer uptake except mild diffuse tracer uptake in the scalp region at the site of the lesion. An MRI Brain revealed multiple melanin deposits in bilateral medial hippocampi and pial surfaces of cerebellum and bilateral parasagittal frontal lobes.
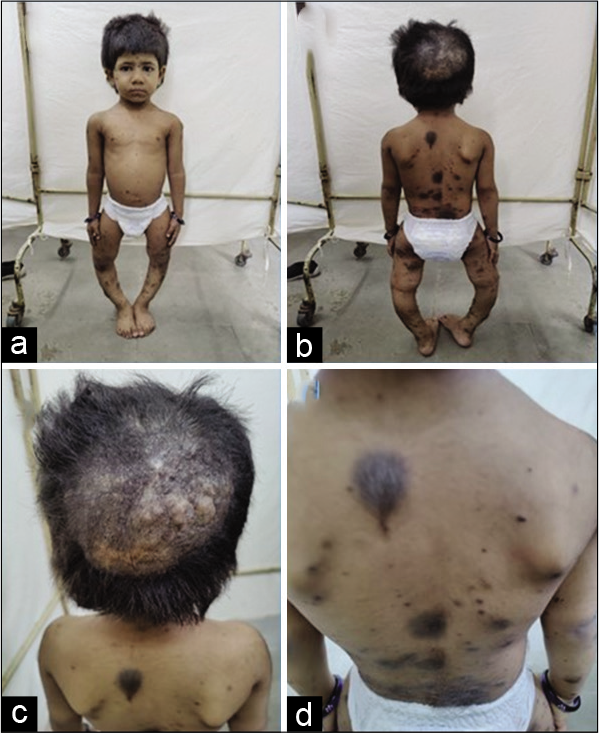
- (a) Short stature with genu varus, (b) Multiple nevi, (c) Verrucous scalp lesion, (d) Hairy pigmented nevi on back.
She was thus diagnosed as congenital neurocutaneous melanotic nevus syndrome with the nevi producing FGF-23 causing hypophosphatemic rickets. She was treated for vitamin D deficiency to which there was a favourable biochemical response and is currently under our follow up.
Cutaneous-skeletal hypophosphatemia syndrome (CHCS) features epidermal or melanocytic nevi and hypophosphatemic rickets with elevated levels of fibroblast growth factor-23 (FGF-23). Affected children often require phosphate and calcitriol supplementation to maintain mineral homeostasis. Convincing data that nevi removal improved blood phosphate level is lacking. Burosumab, a monoconal antibody to FGF23 may have a role in the treatment of this condition.[1] CHCS is a part of the nevus syndrome spectrum which is an example of somatic mosaicism arising via post zygotic activating RAS mutations.[2]
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.





